We’re very happy to welcome new students among us!
😎 We wish all the best to Monalisha Nayak, Giulia Ippoliti, Prasad Shetty and Maru Arias Montecillo in our BµF Lab 






 From left to right:
From left to right:
Ondrej, Ernest, Laura, Nil (Ernest’s and Laura’s boy hidden in the warm blanket), Audrey, Viola, Ayyappa and Saba. We’re expecting a call from Montreal Canadiens any moment 



Sara, Ayyappa, Jamee, Viola, and Audrey (from left to right)
BµF Team

We are pleased to announce that Ayyappasamy Sudalaiyadum Perumal (Ayyappa) just joined our research group as a postdoc!
Welcome to Montreal, Ayyappa!

BµL team 

Who knows what’s next?
Consistently, TTI/Vanguard members who attend [next] do. At our 2015 meeting, it was how to save Moore’s Law; biotechnology losing its separate status and becoming just more technology; and mobility changing everything—even the Internet itself. And that was just the first three speakers (Stanford professor and former Intel principal engineer Subhasish Mitra; DARPA program manager Alicia Jackson; and Benedict Evans, a partner at Andreessen Horowitz). Come to San Francisco and find out what’s next!
December 6–7, 2016, San Francisco
Grand Hyatt San Francisco
345 Stockton Street, San Francisco, California, USA 94108
Prof. Dan V. Nicolau will show how a computer consisting of a nanostructured network can use protein filaments to solve an NP-complete problem (the subset sum problem).
Abstract
Biocomputation and Biosimulation with Molecular-Motors-Propelled Agents
Important and diverse mathematical problems—including cryptography, network routing, and protein folding—require the exploration a large number of candidate solutions. Because the time required for solving these problems grows exponentially with their size, electronic computers, which operate sequentially, cannot solve them in a reasonable timeframe. Unfortunately, the parallel-computation approaches proposed so far—e.g., DNA-, and quantum-computing—suffer from fundamental and practical drawbacks that prevent their successful implementation. Biological entities, from microorganisms to humans, routinely process information in parallel for essential tasks, such as foraging, searching for available space, competition, and cooperation. While several avenues for the use of biological agents for IT tasks exist, the most exciting use a very large number of agents exploring purposefully designed microfluidics networks. For instance, we reported the foundations of a parallel-computation system in which a given combinatorial problem is encoded into a graphical, modular network that is embedded in a nanofabricated planar device. Exploring the network in a parallel fashion using a large number of independent, agents, e.g., molecular motor-propelled agents, then solves the mathematical problem. Besides speed, this approach addresses issues related to power consumption and heat dissipation by using orders-of-magnitude less energy than conventional computers.
Live YouTube stream of [next] 2016 conference:
Day 1
www.youtube.com/watch?v=
Day 2 – Dec 7, 9:10 am (PST)
www.youtube.com/watch?v=
Useful links
List of speakers
Schedule
Accommodation and travel

Our new paper has been just published in Biosensors and Bioelectronics!
Hanson, K. L., Fulga, F., Dobroiu, S., Solana, G., Kašpar, O., Tokárová, V., Nicolau, D. V.
Abstract
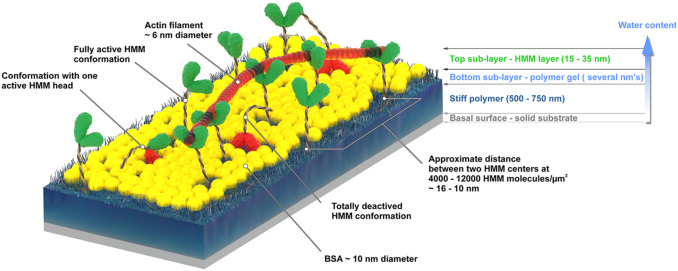 The actin-myosin system, responsible for muscle contraction, is also the force-generating element in dynamic nanodevices operating with surface-immobilized motor proteins. These devices require materials that are amenable to micro- and nano-fabrication, but also preserve the bioactivity of molecular motors. The complexity of the protein-surface systems is greatly amplified by those of the polymer-fluid interface; and of the structure and function of molecular motors, making the study of these interactions critical to the success of molecular motor-based nanodevices. We measured the density of the adsorbed motor protein (heavy meromyosin, HMM) using quartz crystal microbalance; and motor bioactivity with ATPase assay, on a set of model surfaces, i.e., nitrocellulose, polystyrene, poly(methyl methacrylate), and poly(butyl methacrylate), poly(tert-butyl methacrylate). A higher hydrophobicity of the adsorbing material translates in a higher total number of HMM molecules per unit area, but also in a lower uptake of water, and a lower ratio of active per total HMM molecules per unit area. We also measured the motility characteristics of actin filaments on the model surfaces, i.e., velocity, smoothness and deflection of movement, determined via in vitro motility assays. The filament velocities were found to be controlled by the relative number of active HMM per total motors, rather than their absolute surface density. The study allowed the formulation of the general engineering principles for the selection of polymeric materials for the manufacturing of dynamic nanodevices using protein molecular motors.
The actin-myosin system, responsible for muscle contraction, is also the force-generating element in dynamic nanodevices operating with surface-immobilized motor proteins. These devices require materials that are amenable to micro- and nano-fabrication, but also preserve the bioactivity of molecular motors. The complexity of the protein-surface systems is greatly amplified by those of the polymer-fluid interface; and of the structure and function of molecular motors, making the study of these interactions critical to the success of molecular motor-based nanodevices. We measured the density of the adsorbed motor protein (heavy meromyosin, HMM) using quartz crystal microbalance; and motor bioactivity with ATPase assay, on a set of model surfaces, i.e., nitrocellulose, polystyrene, poly(methyl methacrylate), and poly(butyl methacrylate), poly(tert-butyl methacrylate). A higher hydrophobicity of the adsorbing material translates in a higher total number of HMM molecules per unit area, but also in a lower uptake of water, and a lower ratio of active per total HMM molecules per unit area. We also measured the motility characteristics of actin filaments on the model surfaces, i.e., velocity, smoothness and deflection of movement, determined via in vitro motility assays. The filament velocities were found to be controlled by the relative number of active HMM per total motors, rather than their absolute surface density. The study allowed the formulation of the general engineering principles for the selection of polymeric materials for the manufacturing of dynamic nanodevices using protein molecular motors.

Oral presentation
Friday, June 17, 2016 (10:50 – 11:20) – Session X
From Nanodevices to General Theory to Living Cells Heiner Linke, Lund University, Sweden, Chair
Doing Maths with Autonomous Biological Agents
Dan Nicolau, Jr., Molecular Sense, Ltd., United Kingdom
Electronic computers are very good at performing a high number of operations at very high speeds – one-at-a-time. As a result, they struggle with combinatorial tasks that can be only be practically solved if many operations are performed in parallel. This has led to ideas about harnessing the parallelism inherent in biological signal processing for producing a new kind of computing system, such as DNA and molecular computing. In recent work, we presented a proof-of-concept for a parallel molecular-motor driven computer that solves a classic “combinatorial” NP-complete problem – Subset Sum. The device consists of a specifically designed, nanostructured network explored by a large number of molecular-motor-driven, protein filaments. This system is highly energy efficient, avoiding the heating issues limiting electronic computers. In this talk, we will discuss the potential and the challenges involved in developing this kind of technology and how it might be able to tackle problem classes that vex existing computational devices
Posters
Parallel Biocomputational Devices Based on Molecular Motors in Nanostructures
14-POS Board 14
Frida W. Lindberg1 , Till Korten2 , Mercy Lard1 , Mohammad A. Rahman3 , Hideyo Taktsuki3 , Cordula Reuther2 , Falco Van Delft4 , Malin Persson5 , Elina Bengtsson3 , Emelie Haettner1 , Alf Månsson3 , Stefan Diez2 , Dan Jr. V. Nicolau6 , Dan Nicolau7 , Heiner Linke1 .
1 Lund University, Lund, Sweden,
2 Technische Universität Dresden, Dresden, Germany,
3 Linnaeus University, Kalmar, Sweden, 4 High Tech Campus
4, Eindhoven, Netherlands,
5 Karolinska Institutet, Stockholm, Sweden,
6 Molecular Sense Ltd, Oxford, United Kingdom,
7 McGill University, Montreal, QC, Canada.
Solving mathematical problems of a combinatorial nature requires the exploration of a large solution space. As the number of possible solutions grows, this task becomes intractable for traditional, serial computation and therefore, calls for parallel computation techniques. Here we demonstrate an approach to solve a combinatorial problem by parallel computation based on molecular-motor driven biomolecules to explore physical networks of nanoscaled channels in a highly energy-efficient manner (Nicolau et al. 2016). We solve a combinatorial problem known as the subset sum problem, by encoding it into physical networks of channels patterned by lithography. These networks encode binary addition computers. The channel floors are covered with molecular motors that propel protein filaments fed into the network at one end, exploring the network. The filaments’ exit-points correspond to different solutions. Each filament explores one solution, thus, a large number of proteins can be used to compute problems in a massively parallel, energy-efficient manner. We present a proof-of-principle demonstration of the parallel-computation technique, and the status of our ongoing work to optimize and up-scale this system. We test different designs to optimize the individual architectural elements, reducing error rates and increase computing efficiency. We also aim to incorporate switchable junctions into the networks, providing programmable “gates” that can be switched on and off, controlling passage of protein filaments, enabling a high variability of networks. Furthermore, we develop different processing methods for fabricating devices. Our approach is scalable using existing nanofabrication technology. Because one NP complete problem can be converted into another, this technique can be used, in principle, to solve many NP complete problems, with applications in, drug design, scheduling activities, checking of electronic circuit designs, etc. Nicolau, D.V.J. et al., 2016. Massively-parallel computation with molecular motor-propelled agents in nanofabricated networks. PNAS 113(10), pp. 2591–2596.
Fungal Space Searching Can Outperform Standard Algorithms
36-POS Board 36
Hsin-Yu V. Lin, Dan V. Nicolau,
McGill University, Department of Bioengineering, Canada.
The ecological success of basidiomycetous fungi, accounting for ~30% of known fungal species, can be attributed to the efficient expansion of branched filaments (hyphae) when seeking out nutritional resources in the surrounding environment. Despite the fact that these fungi naturally colonize 3D micro-structured media, their growth behaviour has been primarily studied on flat surfaces. Fortunately, microfluidics provides a versatile methodology for the probing the fungal various space search strategies. Solving mazes is a difficult algorithmic exercise, which is why mazes are used to estimate the optimality of the behavioural response, or intelligence, of many higher organisms including ants, bees, mice, rats, octopi, and humans, as well as artificial intelligence-enabled robots. When presented to “intelligence-testing” geometries, e.g., mazes, fungi use a natural program for searching the available space. While different species present different variants of this fungal program, its framework is common and it consists of the interplay of two ‘sub-routines’: collision-induced branching, and directional memory. These studies also demonstrated that the natural program comprising the two ‘sub-routines’ is markedly superior to variants where one of these is, or both are suppressed. A comparison of the performance of the natural algorithm against those of several standard space searching ones revealed that fungi consistently outperforms Depth-First-Search (DFS) algorithm. Although the performance of the natural algorithms is inferior to that of’ informed algorithms’, e.g., A*, this under-performance does not importantly increase with the increase of the size of the maze. These findings encourage a systematic effort to harvest the natural space searching algorithms used by microorganisms, which, if efficient, can be reverse-engineered for graph and tree search strategies

Our article has been just published in the Lab on a Chip!
Abstract
Micro-patterned surfaces with alternate, hydrophilic, and hydrophobic rectangular areas, effectively confine water droplets down to attolitre volumes. The contact angle, volume, and geometry of the confined droplets as a function of the geometry and physico-chemical properties of the confining surfaces have been determined by phenomenological simulations, validated by Atomic Force Microscopy measurements. The combination between experiments and simulations can be used for the purposeful design of surface-addressable hydrophobicity arrays employed in digital microfluidics and high throughput screening nanoarrays.
DOI: 10.1039/C6LC00622A
Accepted 27 May 2016
First published online 27 May 2016

Dan V. Nicolau
Computation with Motile Biological Agents
We are looking for the answer to the Ultimate Question of Life, The Universe, and Everything…